Introduction
Compared to other photovoltaic technologies, perovskite solar cells are currently showing impressive rates of performance improvement. At the time of writing, power conversion efficiencies (PCE) values have reached about 25% with further gains predicted. They are also straightforward to produce and lend themselves to flexible and low-weight devices. Following early key experiments in 2009 and 2012, interest in these devices has exploded and work is now underway to both further optimise their performance and to find viable routes for their commercial application.
What are Perovskite Solar Cells?
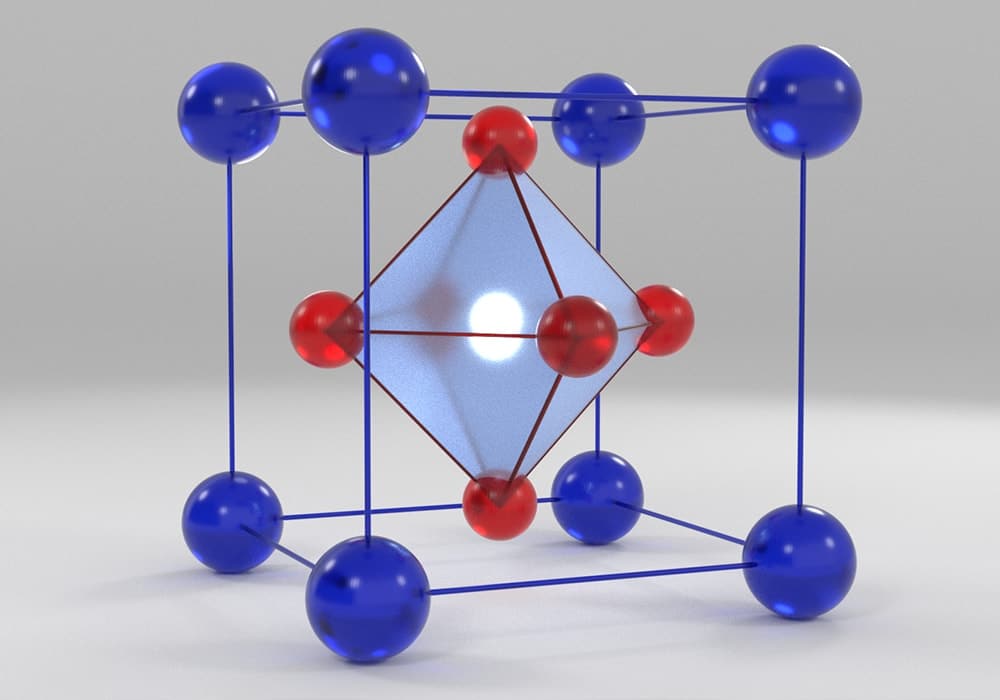
A perovskite solar cell (PSC) is a solar cell incorporating a perovskite material! Definitions of perovskite materials vary, but all require the general formula ABX3, where A and B are cations and X is an anion. Typically, B is a metal cation and X is a halide. Cation A can be organic or inorganic. Importantly, these constituents must be arranged in a specific geometry: BX6 octahedra intercalated with A cations. A visual depiction of this is below:
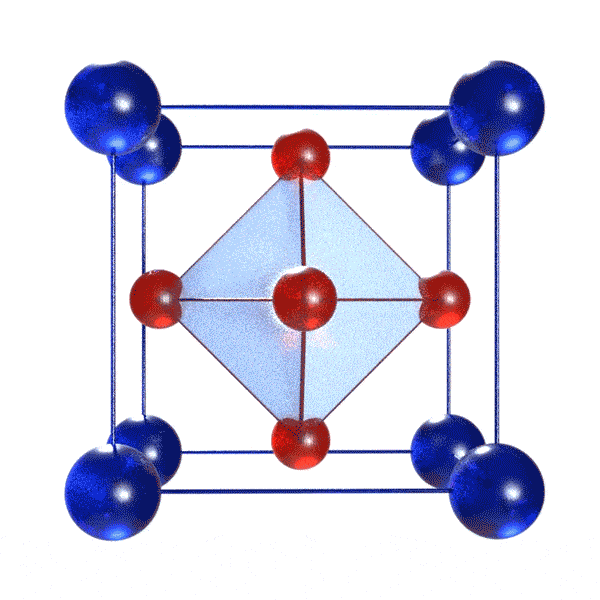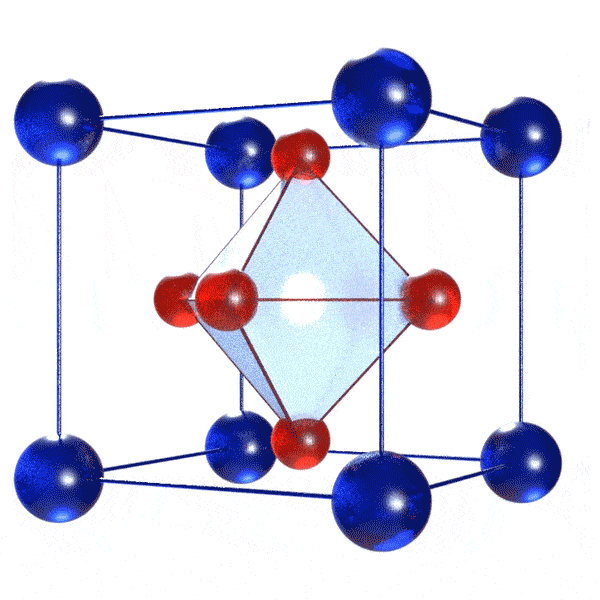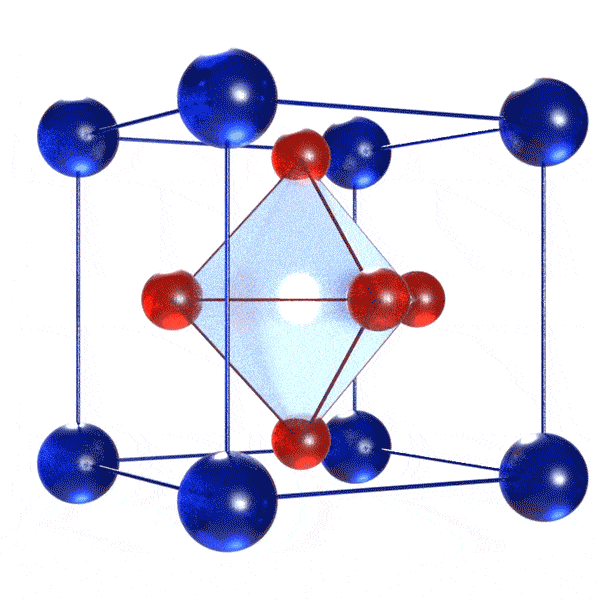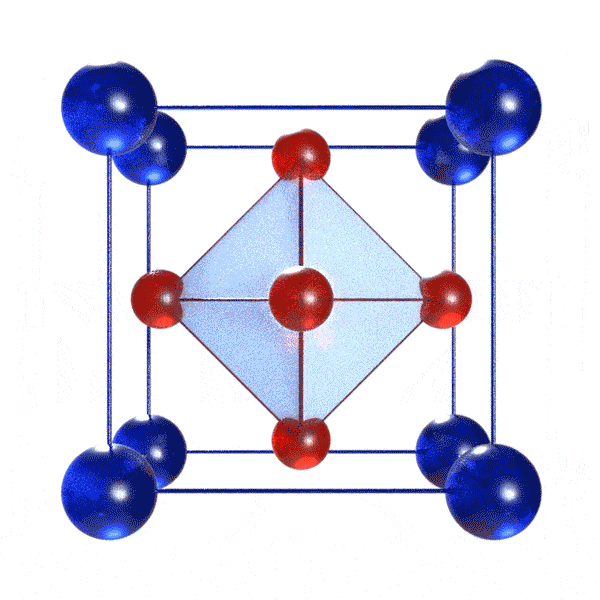
The perovskite material is the light-harvesting substance in PSCs. For this, it absorbs photons that results in electron-hole pair formation. Later, this electron-hole pair may separate (or not, though loss mechanisms underlying solar cell inefficiencies), leading to the release of negative and positive charge carriers. These electron (negative) and hole (positive) charge carriers are collected, separately, by other materials in the device (transport layers) and flow out to generate a voltage in an external circuit.
Various perovskite materials have been investigated for PSCs, with the most common being MAPbI3.This material consists of methylammonium+ cations intercalating an octahedral framework of Pb2+ cations and iodide (I–) anions.
PSC Fabrication

Methods for making PSCs fall into two distinct categories: solution-based and vacuum evaporation.
Solution based approaches, using techniques such as spin-coating, are quick, involve low-cost equipment, give well-performing devices and are quick to establish in the laboratory. However, they are not scalable and therefore poorly-suited to industrial-scale production.
On the other hand, vacuum evaporation (a class of physical vapour deposition—PVD), involves heating perovskite precursors in a vacuum chamber such that they evaporate upwards to coat a substrate. With careful control of the process, the precursors combine to form the required perovskite material. Evaporation approaches also provide excellent results, are compatible with other processes required during solar cell fabrication (e.g., deposition of transport layers and metal contacts—also often done using PVD), and are scalable.
Vacuum Evaporation Systems
To form perovskite materials by vacuum evaporation, precursor materials must be co-evaporated onto a substrate. Co-evaporation means that the two materials must be evaporated simultaneously so that they both arrive at the substrate simultaneously. For example, MAPbI3 requires the co-evaporation of MAI (methylammonium iodide) and PbI (lead iodide). It is essential that the setup allows for the correct balance of deposited materials (that is, they arrive in the correct proportion at the correct rate) to get the desired perovskite structure and properties.

For both MAI and PbI, the evaporation temperatures (approximate temperatures required for evaporation under vacuum) are low as compared to other materials, such as metals, commonly deposited by thermal evaporation. For example, MAI requires up to about only 150 °C, while metal halides such as PbI require up to around 400–500 °C. (Exact temperatures are subject to setup and process.) As such, evaporation sources capable of operating at lower temperatures than standard resistive thermal sources (that are typically geared towards temperatures up to 1800 °C) must be used—without a fine level of control it is very easy to over-apply power leading to an uncontrolled, runaway evaporation (or even the ejection of all material from the source at once).
While evaporation sources for perovskite precursors must be suitable for operating at low temperatures, it is deposition rate that is the critical parameter—and this is not always a straightforward function of reported source temperature. As such, perovskite deposition systems must include in-situ rate monitoring for each precursor source present. Typically, this is done using quartz crystal sensor heads (microbalances) positioned strategically so that each ‘sees’ only the material being evaporated from one source.
For most evaporation processes, material moves in a line-of-sight manner from source to substrate, i.e., once released from the source it travels in a straight line leading to a well-defined evaporation ‘cone’. For example, this is the case when evaporating most metals and allows for ready planning of source-sensor geometries. However, for very volatile materials, including MAI, evaporation leads to a high pressure forming above the source. This pressure can produce sideways movement of material, resulting in unwanted deposition throughout the chamber. As such, for perovskite deposition tools, careful design is needed to prevent ‘cross-talk’ on microbalance measurements due to precursors such as MAI moving freely to coat sensors allocated to different sources.
Moorfield LTE Technology and Systems

Moorfield low-temperature evaporation (LTE) technology allows us to build systems for effective perovskite deposition for PSC work. For these applications, the following features and options are available:
- LTE cells and controllers: Ultra-low thermal mass and optional active water-cooling for precise control and minimal temperature overshoot; temperature, power or deposition rate control, including with PID feedback loop versus sensor measurements
- Quartz crystal sensor heads: Water-cooled for reduced temperature effects, expert design and location to minimise signal ‘cross-talk’ when using high vapour pressure perovskite precursors
- Vacuum system: Expert process chamber design and customisation, including optional corrosion-resistant pumping systems and pre-pump protective features
- Process controllers: State-of-the-art automatic process controllers allowing for multi-stage recipe-based operation, with each stage containing single or multiple sources operating (i.e., co-evaporation) with feedback-loop-controlled rate of each source
. - Multi-technique tools: Allows for the deposition of perovskite plus other PSC layers through different PVD techniques on one system. Furthermore, systems can be fitted with cooling and heating stages for working with sensitive substrates or for annealing during or post-deposition
.
Moorfield tools for PSC work include the benchtop nanoPVD-T15A at the entry-level end, up to fully customisable standalone MiniLab tools where enhanced capability is required.
Glovebox Integration
While finished PSCs can be perfectly useable in ambient conditions, it is often necessary to perform device fabrication under inert atmospheres as moisture and oxygen can prove detrimental to material properties before final protective coatings are applied. To this end, PSC work is commonly carried out within a glovebox containing an inert atmosphere, e.g., pure argon or nitrogen.
Moorfield LTE tools based on our MiniLab 026 and 090 platforms can be integrated with a supporting glovebox allowing for handling of substrates pre- and post-deposition in inert atmospheres. We can work with your preferred glovebox supplier, or suggest one to match your budget and requirements. While our MiniLab 090 tools have box-style process chambers that need a purpose-designed glovebox, our MiniLab 026 systems can be installed in an existing unit if one is already available.
Summary
Perovskite materials show excellent promise for solar cells and vacuum evaporation is a promising method for their fabrication that is also suitable for industrial production. Deposition tools for perovskite work require careful design to allow for controlled production of high-quality material. Moorfield offers the expertise and technology required, including a full range of LTE sources, process control options and complete deposition systems.
Considering a project? Then contact us today to discuss the options.